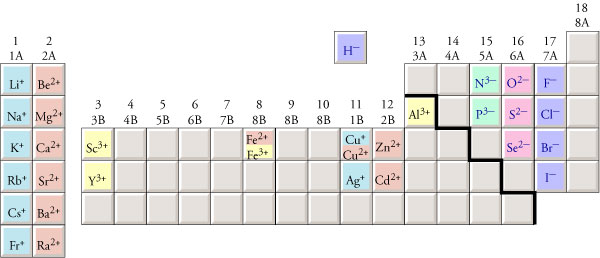
One can use the periodic table to predict whether an. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. Thus, CaCO 3 is named calcium carbonate Na 2SO 4 is named sodium sulfate (NH 4) 2HPO 4 (a compound with two polyatomic ions) is named ammonium hydrogen phosphate and Pb 2+ paired with SO 4 2-, PbSO 4 is named lead (II) sulfate. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. The procedure for naming ionic compounds contain polyatomic ions is identical to that described above for simple ions. That includes BOTH the cation AND the anion involved in the formula. The cation Fe 3+ paired with oxygen would have the formula Fe 2O 3 and would have the name iron (III) oxide. Please remember that all elements involved in this lesson have ONLY ONE charge. Thus Fe 2+ in a compound with chloride would have a formula FeCl 2 and would be named iron (II) chloride, with the oxidation state (the charge on the iron) appearing as a Roman numeral in parenthesis after the cation. The positive cations and the negative anions then attract each other to form ionic bonds. For metals, the oxidation state is the same as the charge. When you are naming an ionic compound containing iron, it is necessary to indicate which oxidation state the metal has. Thus a divalent anion will be formed by a non metal element of group 16 because it will have 6 valence electrons in its atom. Thus, iron exists as Fe 2+ and Fe 3+ cations (they are referred to as “oxidation states”, and will be covered in detail in Chapter 5). A divalent anion is formed by a non-metal atom having 6 valence electrons (so that it can accept 2 more electrons to complete the octet and form a divalent anion). Many transition metals exist as more than one type of cation. There are exceptions to this simple nomenclature, however. Tellurium atom cation: 1: OH-hydroxide anion-1: OH + hydoxyl cation: 1: HS-mercapto anion-1: HS + sulfur monohydride cation: 1: CrH + Chromium hydride cation: 1: HSe-selenium monohydride anion-1: HSe + selenium monohydride cation: 1: H 2 O-water anion-1: H 2 O + water cation: 1: H 2 S-Hydrogen sulfide anion-1: H 2 S + Hydrogen sulfide cation: 1. Then, since the lithium ion has one less electron, remove. For example, to find the configuration for the lithium ion (Li), start with neutral lithium (1s☢s¹). Then, add or remove electrons depending on the ions charge. The chemist reading the name is assumed to have sufficient knowledge to pair the elements properly based on their common valence states. To find the electron configuration for an ion, first identify the configuration for the neutral atom. Some of the examples of divalent anions are oxide (O 2- ), Carbonate (CO 32- ), etc. It is denoted by superscript 2- on the symbol of an atom or group of atoms. This means the number of electrons is higher than protons in divalent anions. For example NaI is named sodium iodide Na 2S is named sodium sulfide CaCl 2 is named calcium chloride. Divalent anions are formed by gaining two valence electrons and attaining a negative charge. When you are constructing names for ionic compounds, you do not use “multipliers” to indicate how many cations or anions are present in the compound. If the anion is a single element, the suffix ide is added to the root name of the element. Nomenclature for these compounds is trivial the cation is named first, followed by the anion. The simplest ionic compounds consist of a single type of cation associated with a single type of anion. Because of their net electrical charge, cations are repelled by other cations and are attracted to anions. An ion may consist of a single atom of an element (a monatomic ion or monatomic cation or anion) or of several atoms that are bonded together (a polyatomic ion or polyatomic cation or anion). Note: The transition metal is underlined in the following compounds.\) A cation has fewer electrons than protons. 
Anions are negatively charged ion and are usually found last in a chemical formula. The number of elements retained as seen in this picture represents the cation exchange capacity of this peat particle. The peat particle pictured has negative charges that have cations attached to these sites. Since the 3p orbitals are all paired, this complex is diamagnetic.ĭetermine the oxidation states of the transition metals found in these neutral compounds. Cations are positively charged ions and are usually found first in a chemical formula. In comparison, the cation exchange capacity is more significant in a soilless media than the anion exchange capacity. \) is potassium permanganate, where manganese is in the +7 state with no electrons in the 4s and 3d orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
